The Technology
The heart of our solution is an analyser that uses lasers to verify the identity and concentration of IV drugs. The components of the service include the analyser, delivery consumables, a cloud-based library of drug data, and anonymised patient data. Our first market application is the verification of oncology drugs in compounding pharmacies.

Current Desktop Consumable Analyser

SMART CELL
Veriphi’s fourth-generation analyser (the CAV4) is our latest model, featuring onboard controls, reduced cycle times, and enhanced sensitivity to verify IV drug identity and concentration via a proprietary connector.
The vision for the next-generation product is a miniaturised analyser that can be used at the patient’s bedside, in hospital or community settings, including remote areas.
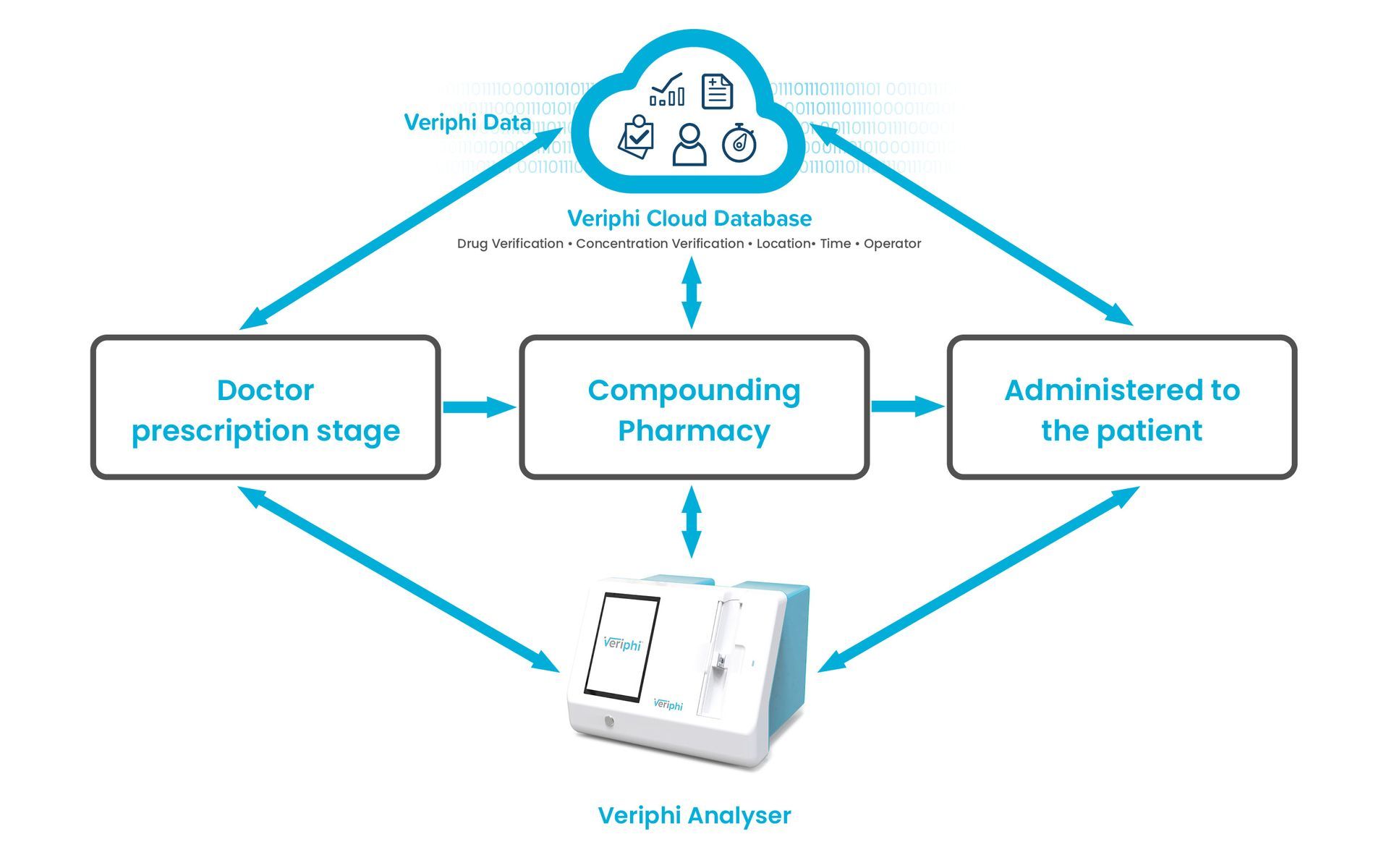
Our analyser is the keystone to a hardware-enabled, cloud-based medication management solution. Veriphi’s bench-top analyser is our first commercial solution and uses lasers to recognise the unique spectral signature for each IV drug compound at specific concentrations. At the same time, our software compares the recorded drug signature with a known library of drugs to determine whether the drug is what the clinician intended. If not, the analyser raises an alarm to protect the patient. Veriphi’s solution can reduce injury, isolate problem areas and create greater transparency for IV medication delivery.
Our analyser is the keystone to a hardware-enabled, cloud-based medication management solution. Veriphi’s bench-top analyser is our first commercial solution and uses lasers to recognise the unique spectral signature for each IV drug compound at specific concentrations. At the same time, our software compares the recorded drug signature with a known library of drugs to determine whether the drug is what the clinician intended. If not, the analyser raises an alarm to protect the patient. Veriphi’s solution can reduce injury, isolate problem areas and create greater transparency for IV medication delivery.
The Problem
Globally, the cost associated with medication errors has been estimated to be US$42 billion per year.¹
The Global Trend - Hospital At Home
Veriphi’s goal is to enable safe and effective administration of oncology drugs to cancer patients wherever they might be as the world moves from hospital-centric to patient-centric models of care. A catalyst for change, the COVID-19 pandemic has led to a review of hospital treatment of patients, both for those with the virus and those typically requiring hospital treatment. The ‘hospital at home’ model has been expedited worldwide and in recent years, a handful of US systems have launched such programmes. These join already firmly established models in the UK, Australia, Canada, Israel and other countries with government-run health systems.²
Focus on Oncology
Veriphi’s initial focus is IV oncology treatments because we believe this area will provide the greatest opportunity to improve patient outcomes. Intravenous errors are twice as likely to harm patients than drugs administered by other means,³ and chemotherapy errors occur at a rate of about one to four per 1,000 orders and affect at least 1 to 3% of adult and paediatric oncology patients.⁴ The consequences of oncology errors can have a more significant impact than other medications.
Sustainable Competitive Advantage
A comprehensive global patent portfolio protects our first-mover advantage with eight US patents granted.
¹ WHO, Global Patient Safety Challenge: Medication Without Harm 2017.
² Transforming care, Has the Time Finally Come for Hospital at Home? The Commonwealth Fund, July 2020.
³ American Journal of Health-System Pharmacy 2008.
⁴ Weingart N et al. Chemotherapy Medication Errors, The Lancet, 2018.
PATENTS
• 8 granted US patents
• 22 granted global patents
PARTNERS
The University of Auckland, School of Pharmacy, School of Chemistry and School of Medical Science; Dodd-Walls Centre for Photonic and Quantum Technologies
COMPLIANCE
International Electrotechnical
Commission (IEC) approvals pending

